|
Biodiversity Data Journal :
Taxonomic paper
|
Chromosome studies in the aquatic monocots of Myanmar: A brief review with additional records
|
Corresponding author:
Academic editor: Lorenzo Peruzzi
Received: 11 Feb 2014 | Accepted: 08 May 2014 | Published: 13 May 2014
© 2014 Yu Ito, Nobuyuki Tanaka
This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation:
Ito Y, Tanaka N (2014) Chromosome studies in the aquatic monocots of Myanmar: A brief review with additional records. Biodiversity Data Journal 2: e1069. https://doi.org/10.3897/BDJ.2.e1069
|
|
Abstract
Myanmar (Burma) constitutes a significant component of the Indo-Myanmar biodiversity hotspot, with elements of the Indian, the Indochina, and the Sino-Japanese floristic regions, yet thus far only a few reliable sources of the country's flora have been available. As a part of a contribution for the floristic inventory of Myanmar, since it is important in a floristic survey to obtain as much information as possible, in addition to previous two reports, here we present three more chromosome counts in the aquatic monocots of Myanmar: Limnocharis flava with 2n = 20, Sagittaria trifolia with 2n = 22 (Alismataceae), and Potamogeton distinctus × P. nodosus with 2n = 52 (Potamogetonaceae); the third one is new to science. A brief review of cytological researches in the floristic regions' 45 non-hybrid aquatic monocots plus well investigated two inter-specific hybrids that are recorded in Myanmar is given, indicating that the further works with a focus on species in Myanmar that has infra-specific chromosome variation in the floristic regions will address the precise evolutionary history of the aquatic flora of Myanmar.
Keywords
Aquatic plants, chromosome counts, Limnocharis, Myanmar, Potamogeton, Sagittaria
Introduction
With its wealth of plant diversity, Myanmar (Burma) constitutes a significant component of the Indo-Myanmar biodiversity hotspot with elements of the India, the Indochina, and the Sino-Japanese floristic regions (ca, 13,500 vascular plants:
The aim of floristic research is not only to count the total number of species but also to evaluate the native flora’s evolutionary origins by comparing with related floristic regions. From this aspect, it is useful to obtain as much information as possible, e.g., chromosome data (
Aquatic plants, which is polyphyletically evolved in fern and fern allies, basal angiosperms, monocots, and eudicots, is known as having numerous chromosomal variation, thus an excellent model for this aim. Here, in addition to the previous contributions of chromosome counts for new or noteworthy aquatic plants from Myanmar (Najas tenuis:
Materials and methods
Chromosome observation
Plant materials of Limnocharis flava (Alismataceae), Sagittaria trifolia (Alismataceae), Najas tenuis (Hydrocharitaceae), Nechamandra alternifolia (Hydrocharitaceae), and Potamogeton distinctus × P. nodosus (Potamogetonaceae) were collected in the expeditions to Myanmar (Bago Division and Shan State) in 2008. The collections were rigorously identified based on morphological characters using the original protologues as well as a previous taxonomic treatment by
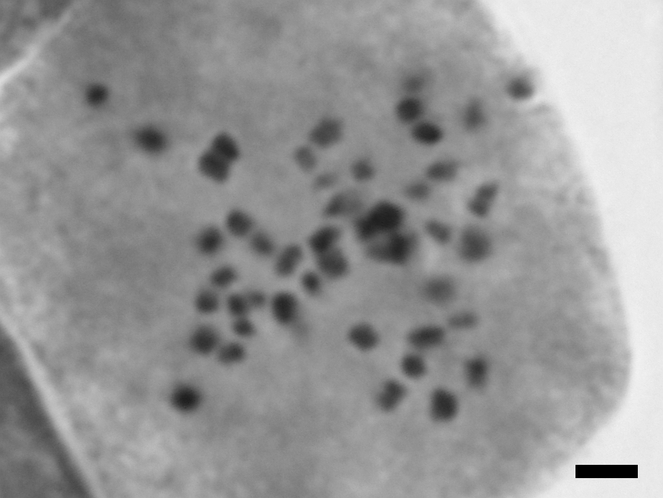
Root tips collected in the field were pretreated with 0.002 M 8-hydroxyquinoline at 4 °C in 12 h, and fixed with freshly mixed Carnoy’s fixative (3: 1 ethyl alcohol: acetic acid) for at least 30 min, and then preserved at 4 °C in 12 h. For microscopic observation, root tips were soaked in 1 N HCl for 1 h followed by 10 min at 60 °C. After being immersed in tap water, the materials were stained in a drop of 1.5% orcein acetate solution on a slide glass in 5 min., and then squashed. Then somatic chromosome numbers of the three taxa were obtained by light microscopic examination. For each species, at least two cells were used to confirm the numbers.
Distribution for each species follows
Literature review
Chromosome researches for aquatic monocots of Myanmar were reviewed with a broad focus on Myanmar and related floristic regions, i.e., the Indian, the Indochina, and the Sino-Japanes floristic regions. The focal species include 45 non-hybrid aquatic monocots listed in
Chromosome counts for the aquatic monocots of Myanmar
Order
Family
Genus Limnocharis
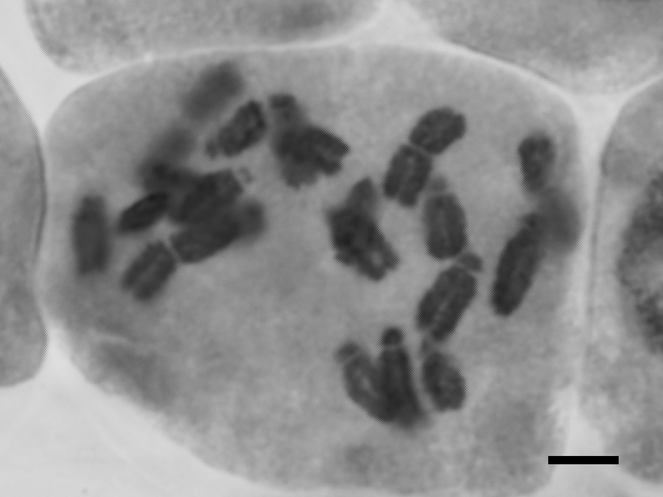
Limnocharis flava
-
country: Myanmar; stateProvince:Bago; municipality:Pyat Township; locality:along the roadside, paddy field, ca. 30 km east of Pyat; verbatimLatitude:18°49'44"N; verbatimLongitude:95°18'06"E; eventDate:7 Dec 2008; recordedBy:Y. Ito; collectionID:N. Tanaka & al. 080776; institutionCode:MBK, RAF, TI
Native to Americas; naturalized to tropical Asia.
Genus Sagittaria
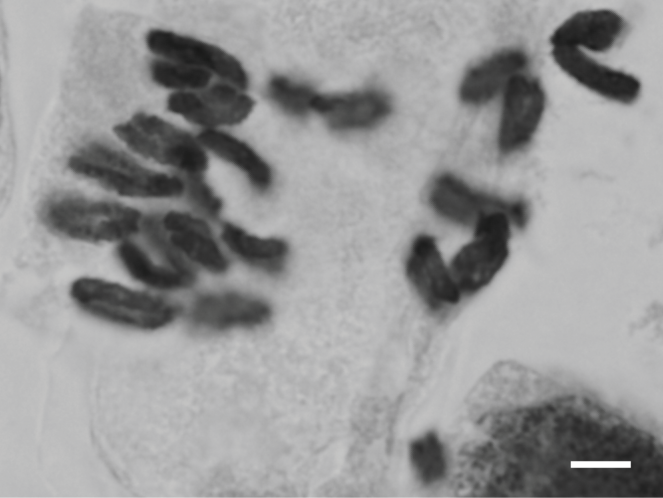
Sagittaria trifolia
-
country: Myanmar; stateProvince:Shan; verbatimLocality:Pindaya; verbatimLatitude:20°59'57"N; verbatimLongitude:96°39'59"E; eventDate:1 Dec 2008; recordedBy:Y. Ito; collectionID:N. Tanaka & al. 080623
Bangladesh, Bhutan, China (nationwide), India (nationwide), Indonesia (Borneo, Java, Sulawesi), Japan, Malaysia (Peninsular), Myanmar, Nepal, Pakistan, Philippines, Thailand; Oceania.
Family
Genus Najas
Najas tenuis
-
country: Myanmar; stateProvince:Shan; verbatimLocality:Inlay Lake, Nyaung Shwe Township; verbatimLatitude:20°32'02"N; verbatimLongitude:96°53'53"E; eventDate:3 Dec 2008; recordedBy:Y. Ito; collectionID:N. Tanaka & al. 080642; institutionCode:MBK, RAF, TI
India (Central, Southern), Myanmar, Sri Lanka.
Genus Nechamandra
Nechamandra alternifolia
-
country: Myanmar; stateProvince:Shan; verbatimLocality:Near Yae Aye Kan Dam, Yae Aye Kan, Kalaw Township; verbatimLatitude:20°35'37"N; verbatimLongitude:96°31'46"E; eventDate:26 Nov 2008; recordedBy:Y. Ito; collectionID:N. Tanaka & al. 080058; institutionCode:MBK, RAF, TI
Bangladesh, China (Southern), India (Eastern, Northern, Southern), Myanmar, Nepal, Sri Lanka, Thailand, Vietnam; Yemen, and Sudan.
Family
Genus Potamogeton
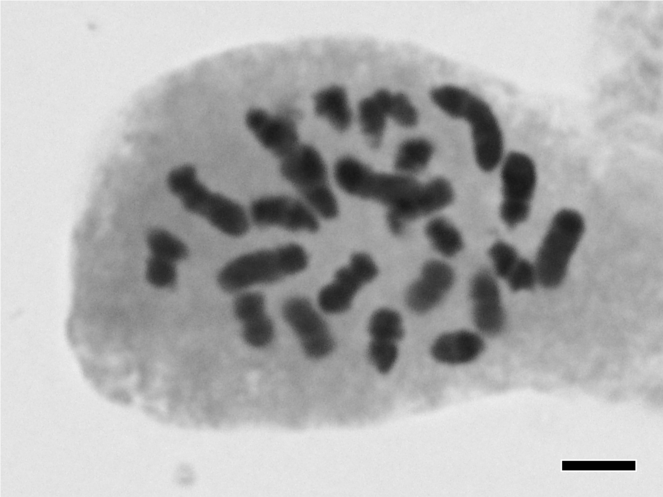
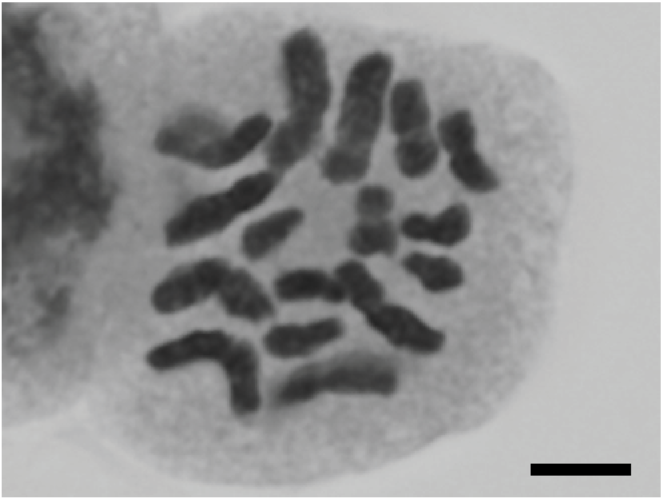
Potamogeton distinctus A. Benn. × P. nodosus Poir.
-
country: Myanmar; stateProvince:Shan; verbatimLocality:Inle Lake; verbatimLatitude:20°27'28"N; verbatimLongitude:96°50'37"E; eventDate:4 Dec 2008; recordedBy:Y. Ito; collectionID:N. Tanaka & al. 080662; institutionCode:MBK, RAF, TI
Analysis
The chromosome counts given for 45 non-hybrid species of aquatic monocots of Myanmar as well as well-investigated two Potamogeton hybrids among them were reviewed with a focus on infra-specific chromosome variation (Table
The chromosome counts given for 45 non-hybrid species of aquatic monocots of Myanmar as well as well-investigated two Potamogeton hybrids among them. Those recorded from neighboring regions are also provided. The species that have no chromosome counts anywhere in the world are shown with n/a. For some species, mostly cosmopolitan ones, only a few representative literature references are given for each chromosome number. Note that due to incapability of original references, some rare chromosome counts are not included in this table: 2n = 18, 42, 48 for Acorus calamus; 2n = 18, 22 for Acorus gramineus (Acoraceae), 2n = 28 for Pistia stratiotes var. cuneata Engl.; 2n = 28 for Pistia stratiotes var. spathulata (Michx.) Engl.; 2n = 20, 50, 60, 80 for Lemna aequinoctialis; 2n = 44 for Lemna trisulca; 2n = 30, 50 for Spirodela polyrrhiza (Araceae); n = 14 (2n = 28), 2n = 10, 12 for Alisma plantago-aquatica; 2n = 22 for Caldesia parnassifolia; 2n = 26, 39 for Limnocharis flava; 2n = 22 for Sagittaria trifolia var. longiloba (Turr.) Mak.; 2n = 22 for Sagittaria trifolia var. sinensis Sims; 2n = 22 for Sagittaria trifolia var. edulis (Sieb.) Ohwi (Alismataceae); 2n = 24 for Blyxa aubertii; 2n = 60 for Najas marina; 2n = 12+1B for Najas marina var. intermedia (Gorski) A. Braun; 2n = 22, 52, 72, 88, 132 for Ottelia alismoides; 2n = 16, 22, 28, 33 for Vallisneria spiralis (Hydrocharitaceae); 2n = 64 for Eichhornia crassipes; 2n = 26, n = 40 (2n = 80) for Monochoria vaginalis (Pontederiaceae); 2n = 60 for Typha angustifolia (Typhaceae). Also refer to previous cytological reviews (aquatic plants:
| Order | Family | Species | Chromo-some number | Floristic region | ||||
|---|---|---|---|---|---|---|---|---|
| Indian | Myanmar | Indo-china | Sino-Japanese | Others | ||||
| Acorales | Acoraceae | Acorus calamus L. | 2n = 24 | |||||
| Acorales | Acoraceae | Acorus calamus L. | 2n = 35 | |||||
| Acorales | Acoraceae | Acorus calamus L. | 2n = 36 | |||||
| Acorales | Acoraceae | Acorus calamus L. | 2n = 44 | |||||
| Acorales | Acoraceae | Acorus calamus L. | 2n = 45 | |||||
| Acorales | Acoraceae | Acorus calamus L. | 2n = 66 | |||||
| Acorales | Acoraceae | Acorus gramineus Sol. ex Aiton | 2n = 24 | |||||
| Acorales | Araceae | Cryptocoryne crispatula Engl. | 2n = 36 | |||||
| Acorales | Araceae | Cryptocoryne crispatula Engl. | 2n = 54 | |||||
| Acorales | Araceae | Cryptocoryne cruddasiana Prain | n/a | |||||
| Acorales | Araceae | Pistia stratiotes L. | 2n = 28 | |||||
| Acorales | Araceae | Landoltia punctata (G. Mey.) Les & D.J. Crawford | n/a | |||||
| Acorales | Araceae | Lemna aequinoctialis Welw. | 2n = 40 | |||||
| Acorales | Araceae | Lemna trisulca L. | 2n = 20 | |||||
| Acorales | Araceae | Lemna trisulca L. | 2n = 40 | |||||
| Acorales | Araceae | Lemna trisulca L. | 2n = 60 | |||||
| Acorales | Araceae | Lemna trisulca L. | 2n = 80 | |||||
| Acorales | Araceae | Spirodela polyrrhiza (L.) Schleid. | 2n = 40 | |||||
| Acorales | Araceae | Spirodela polyrrhiza (L.) Schleid. | 2n = 42 | |||||
| Acorales | Araceae | Spirodela polyrrhiza (L.) Schleid. | 2n = 80 | |||||
| Acorales | Araceae | Wolffia globosa (Roxb.) Hartog &Plas | n/a | |||||
| Alismatales | Alismataceae | Alisma plantago-aquatica L. | 2n = 14 | |||||
| Alismatales | Alismataceae | Caldesia parnassifolia (Bassi ex L.) Parl. | n/a | |||||
| Alismatales | Alismataceae | Limnocharis flava (L.) Buchenau | 2n = 20 | This study | ||||
| Alismatales | Alismataceae | Sagittaria trifolia L. | 2n = 22 | This study | ||||
| Alismatales | Hydrocharita | Blyxa aubertii Rich. | 2n = 40 | |||||
| Alismatales | Hydrocharita | Blyxa echinosperma (C.B. Clarke) Hook. f. | 2n = 42 | |||||
| Alismatales | Hydrocharita | Blyxa echinosperma (C.B. Clarke) Hook. f. | 2n = 74 | |||||
| Alismatales | Hydrocharita | Blyxa japonica (Miq.) Maxim. ex Asch. & Gürke | 2n = 42 | |||||
| Alismatales | Hydrocharita | Blyxa japonica (Miq.) Maxim. ex Asch. & Gürke | 2n = 72 | |||||
| Alismatales | Hydrocharita | Egeria densa (Planch.) Casp. | 2n = 46 | |||||
| Alismatales | Hydrocharita | Egeria densa (Planch.) Casp. | 2n = 48 | |||||
| Alismatales | Hydrocharita | Elodea nuttallii (Planch.) H. St. John | 2n = 48 | |||||
| Alismatales | Hydrocharita | Hydrilla verticillata (L. f.) Royle | 2n = 16 | |||||
| Alismatales | Hydrocharita | Hydrilla verticillata (L. f.) Royle | 2n = 24 | |||||
| Alismatales | Hydrocharita | Hydrilla verticillata (L. f.) Royle | 2n = 32 | |||||
| Alismatales | Hydrocharita | Hydrocharis dubia (Blume) Backer | 2n = 16 | |||||
| Alismatales | Hydrocharita | Najas graminea Delile | 2n = 12 | |||||
| Alismatales | Hydrocharita | Najas graminea Delile | 2n = 24 | |||||
| Alismatales | Hydrocharita | Najas graminea Delile | 2n = 36 | |||||
| Alismatales | Hydrocharita | Najas indica (Willd.) Cham. | n/a | |||||
| Alismatales | Hydrocharita | Najas marina L. | 2n = 12 | |||||
| Alismatales | Hydrocharita | Najas marina L. | 2n = 24 | |||||
| Alismatales | Hydrocharita | Najas tenuis Magnus | 2n = 24 | |||||
| Alismatales | Hydrocharita | Nechamandra alternifolia (Roxb.) Thwaites | 2n = 16 | |||||
| Alismatales | Hydrocharita | Ottelia alismoides (L.) Pers. | 2n = 44 | |||||
| Alismatales | Hydrocharita | Ottelia alismoides (L.) Pers. | 2n = 66 | |||||
| Alismatales | Hydrocharita | Ottelia alismoides (L.) Pers. | 2n = 68 | |||||
| Alismatales | Hydrocharita | Ottelia cordata (Wall.) Dandy | n/a | |||||
| Alismatales | Hydrocharita | Vallisneria spiralis L. | 2n = 20 | |||||
| Alismatales | Hydrocharita | Vallisneria spiralis L. | 2n = 24 | |||||
| Alismatales | Hydrocharita | Vallisneria spiralis L. | 2n = 30 | |||||
| Alismatales | Hydrocharita | Vallisneria spiralis L. | 2n = 40 | |||||
| Alismatales | Aponogetona | Aponogeton lakhonensis A. Camus | n/a | |||||
| Alismatales | Potamogetona | Potamogeton crispus L. | 2n = 52 | |||||
| Alismatales | Potamogetona | Potamogeton crispus L. | 2n = 56 | |||||
| Alismatales | Potamogetona | Potamogeton distinctus A. Benn. | 2n = 52 | |||||
| Alismatales | Potamogetona | Potamogeton distinctus A. Benn. × P. nodosus Poir. | 2n = 52 | This study | ||||
| Alismatales | Potamogetona | Potamogeton maackianus A Benn. | 2n = 52 | |||||
| Alismatales | Potamogetona | Potamogeton maackianus A Benn. | 2n = 56 | |||||
| Alismatales | Potamogetona | Potamogeton × malainoides Miki | 2n = 52 | |||||
| Alismatales | Potamogetona | Potamogeton lucens L. | 2n = 52 | |||||
| Alismatales | Potamogetona | Potamogeton nodosus Poir. | 2n = 52 | |||||
| Alismatales | Potamogetona | Potamogeton octandrus Poir. | 2n = 28 | |||||
| Alismatales | Potamogetona | Potamogeton wrightii Morong | 2n = 52 | |||||
| Alismatales | Potamogetona | Stuckenia pectinata (L.) Börner | 2n = 78 | |||||
| Alismatales | Potamogetona | Stuckenia pectinata (L.) Börner | 2n = 84 | |||||
| Alismatales | Ruppiaceae | Ruppia maritima L. | 2n = 20 | |||||
| Ruppia maritima L. | 2n = 40 | |||||||
| Asparagales | Amaryllidaceae | Crinum thaianum J. Schul. | n/a | |||||
| Commelinales | Pontederiaceae | Eichhornia crassipes (Mart.) Solms | 2n = 32 | |||||
| Commelinales | Pontederiaceae | Monochoria hastata (L.) Solms | 2n = 28 | |||||
| Commelinales | Pontederiaceae | Monochoria hastata (L.) Solms | 2n = 80 | |||||
| Commelinales | Pontederiaceae | Monochoria vaginalis (Burm.f.) C. Presl ex Kunth | 2n = 24 | |||||
| Commelinales | Pontederiaceae | Monochoria vaginalis (Burm.f.) C. Presl ex Kunth | 2n = 48 | |||||
| Commelinales | Pontederiaceae | Monochoria vaginalis (Burm.f.) C. Presl ex Kunth | 2n = 52 | |||||
| Commelinales | Typhaceae | Typha angustifolia L. | 2n = 30 | |||||
| Poales | Eriocaulaceae | Eriocaulon setaceum L. | n/a | |||||
Discussion
Of 45 non-hybrid aquatic monocots and two interspecific hybrids among them, more than two thirds have no chromosome variation. Meanwhile, the following nine species have infra-specific chromosome variation, i.e., Acorus calamus, Cryptocoryne crispatula, Blyxa echinosperma, Hydrilla verticillata, Najas graminea, Ottelia alismoides, Vallisneria spiralis, Monochoria hastata, and M. vaginalis (Table
Potamogeton is known as having numerous inter-specific hybrids, and each parental combination is varied from intra-ploidy crosses to inter-ploidy ones (
Acknowledgements
Thanks are due to the following personnel in Myanmar for their help in arranging our field work: U. Htun Paw Oo, Ex-director of Nature and Wildlife Conservation Division; U. Soe Win Hlaing, Ex-director General, Forest Department, Ministry of Environmental Conservation and Forestry. We also thank Prof. T. Koyama, director of the Kochi Prefectural Makino Botanical Garden (Japan), who initiated the Myanmar-Japanese cooperative program to inventory the plants of Myanmar, and gave us the opportunities to study the Myanmar plants, Prof. J. Murata, curator of the University of Tokyo (Japan), who principally organizes the Myanmar-Japanese inventory project, and Dr. T. Ohi-Toma, assistant professor of the University of Tokyo (Japan), who helps the first author to use the facilities of the botanical gardens, the university of Tokyo.
Author contributions
Conceived and designed the study: YI NT. Collected the samples in the field: YI NT. Performed the chromosome observation: YI. Analyzed the data: YI. Wrote the paper: YI. Corrected and revised manuscript: NT.
References
- Contribution to a cytological catalogue of the British and Irish flora 3.Watsonia19:269‑271. URL: http://archive.bsbi.org.uk/Wats19p269.pdf
- Chromosome numbers and taxonomy in Cryptocoryne (Araceae). II.Nordic Journal of Botany2(5):453‑463. https://doi.org/10.1111/j.1756-1051.1982.tb01208.x
- A revision of Lemna paucicostata group in Japan.Acta Phytotaxonomica et Geobotanica36:45‑58. URL: http://ci.nii.ac.jp/els/110003760235.pdf?id=ART0004972021&type=pdf&lang=jp&host=cinii&order_no=&ppv_type=0&lang_sw=&no=1396668638&cp=
- Cytological studies on three aquatic members of Hydrocharitaceae in relation to their morphological and ecological characteristics.Cytologia43(1):1‑19. https://doi.org/10.1508/cytologia.43.1
- IAPT/IOPB chromosome data 6.Taxon57:1267‑1268.
- Cytology of Monochoria vaginalis complex Presl.Cytologia48(3):627‑631. https://doi.org/10.1508/cytologia.48.627
- Aquatic and Wetland Plants of India: A reference book and identification manual for the vascular plants found in permanent or seasonal fresh water in the subcontinent of India south of the Himalayas.Oxford University Press,Oxford,385pp.
- Chromosome numbers of miscellaneous angiosperms.Annals of the Missouri Botanical Garden68(1):222. https://doi.org/10.2307/2398824
- Chromosome Numbers of Flowering Plants.Academy of Sciences of USSR,Leningrad,926pp.
- Chromosomal studies on Neotropical Limnocharitaceae (Alismatales).Aquatic Botany74(1):33‑41. https://doi.org/10.1016/s0304-3770(02)00038-4
- Cytochemical heterochromatin differentiation in Sinapis alba (Cruciferae) using a simple air-drying technique for producing chromosome spreads.Plant Systematics and Evolution158:97‑106. https://doi.org/10.1007/bf00936336
- Cytological studies in Helobiae: I. Chromosome idiograms and a list of chromosome numbers in seven families.Cytologia21(3):306‑328. https://doi.org/10.1508/cytologia.21.306
- An updated checklist of aquatic plants of Myanmar and Thailand.Biodiversity Data Journal2:e1019. https://doi.org/10.3897/bdj.2.e1019
- Hybridization and polyploidy of an aquatic plant, Ruppia (Ruppiaceae), inferred from plastid and nuclear DNA phylogenies.American Journal of Botany97(7):1156‑1167. https://doi.org/10.3732/ajb.0900168
- New or noteworthy plant collections from Myanmar (3) Caldesia parnassifolia, Nechamandra alternifolia, Potamogeton maackianus and P. octandrus.The Journal of Japanese Botany84(6):321‑329.
- DNA barcoding reveals a new record of Potamogeton distinctus (Potamogetonaceae) and its natural hybrids, P. distinctus × P. nodosus and P. distinctus × P. wrightii (P. × malainoides) from Myanmar.Biodiversity Data Journal2:e1073. https://doi.org/10.3897/bdj.2.e1073
- New or noteworthy plant collections from Myanmar (8) Blyxa aubertii var. echinosperma, Lemna trisulca, and Najas tenuis.APG: Acta Phytotaxonomica et Geobotanica65(2):xx‑xx.
- Chromosome numbers and taxonomy in Cryptocoryne (Araceae).Botaniska Notiser130:71‑87.
- Revision of chromosome numbers of Potamogetonaceae: a new basis for taxonomic and evolutionary implications.Preslia85:421‑487. URL: http://www.preslia.cz/P134Kaplan.pdf
- Chromosome numbers in selected monocotyledons (Czech Republic, Hungary, and Slovakia).Preslia75:97‑113. URL: http://www.preslia.cz/P032CKra.pdf
- A Checklist of the Trees, Shrubs, Herbs, and Climbers of Union of Myanmar.Contributions from the United States National Herbarium. National Museum of Natural History, Washington D.C.,1-590pp.
- Chromosome morphology and number in various populations of Hydrilla verticillata (L.f.) Royle.Aquatic Botany42(3):253‑263. https://doi.org/10.1016/0304-3770(92)90026-f
- Studies of hybridization and chromosome number variation in aquatic angiosperms: evolutionary implications.Aquatic Botany44:181‑228. https://doi.org/10.1016/0304-3770(93)90071-4
- Chromosome numbers of central and northwest European plant species.Opera Botanica5:1‑581.
- Chromosome number reports LXXII.Taxon30:699‑701.
- Cytological studies of some helobiales of Kashmir Himalayas. I. Family Alismataceae.Cytologia49(2):295‑304. https://doi.org/10.1508/cytologia.49.295
- Chromosome numbers of some aquatic plants collected in Toyama Prefecture.Toyama-no-Seibutsu37:1‑6. [InJapanese].
- The distribution and status of Acorus (Araceae) in Canada.Canadian Journal of Botany62(11):2248‑2252. https://doi.org/10.1139/b84-305
- Cytological studies of some helobiales of Kashmir Himalayas. II. Families: Butomaceae and Hydrocharitaceae.Cytologia49(2):305‑312. https://doi.org/10.1508/cytologia.49.305
- Polyploidy in Monochoria hastata Solms, and M. vaginalis Prest. grown in Bangladesh.Cytologia54(3):505‑511. https://doi.org/10.1508/cytologia.54.505
- Citogenética de angiospermas coletadas em Pernambuco - V.Acta Botanica Brasilica13(1):49‑60. [InPortuguese]. URL: http://www.scielo.br/pdf/abb/v13n1/v13n1a06.pdf
- Cytological studies on south Indian araceae.Cytologia43(2):289‑303. https://doi.org/10.1508/cytologia.43.289
- Chromosome numbers from the flora of the Juan Fernandez islands.American Journal of Botany70(6):799‑810. https://doi.org/10.2307/2442929
- Chromosome number reports LXVII.Taxon29:360‑361.
- Cytotaxonomy of Helobiae with special reference to the mode of evolution.Cytologia32(2):286‑307. https://doi.org/10.1508/cytologia.32.286
- Taxonomy of Elodea Michx in the British Isles.Watsonia16:1‑14. URL: http://archive.bsbi.org.uk/Wats16p1.pdf
- Cytology and cytogenetics as a fundamental taxonomic resource for the 20th and 21st centuries.Taxon49(3):451. https://doi.org/10.2307/1224344
- Cytotaxonomical studies in South Indian Araceae.Cytologia53(1):59‑66. https://doi.org/10.1508/cytologia.53.59
- Chromosome numbers and a new model for karyotype evolution in Ruppia L. (Ruppiaceae).Aquatic Botany45(1):1‑13. https://doi.org/10.1016/0304-3770(93)90048-2
- Plant inventory research: contributions to the flora of Myanmar.APG: Acta Phytotaxonomica et Geobotanica56:1‑26. URL: http://ci.nii.ac.jp/naid/110006318373
- Plant inventory research in Myanmar.Bunrui10(2):139‑149. [InJapanese]. URL: http://ci.nii.ac.jp/naid/110007681490
- The flowering plants of Mt. Popa, central of Myanmar - Results of Myanmar-Japanese joint expeditions 2000-2004.Makinoa New Series5:1‑95.
- Karyomorphological studies on some taxa of the Helobiae.Journal of Science, Hiroshima University, Series B, Division 2 (Botany)22:271‑352.
- Cytological variation within Lemna L.Aquatic Botany1:377‑394. https://doi.org/10.1016/0304-3770(75)90038-8
- Van Dijk PP, Tordoff AW, Fellowes J, Lau M, Jinshuang M (2004) Indo-Burma. In: Mittermeier RA, Robles GP, Hoffmann M, Lamoreaux J, da Fonseca GA (Eds) Hotspots Revisited: Earth’s Biologically Richest and Most Endangered Terrestrial Ecoregions.CEMEX, Agrupación Sierra Madre,323-330pp.
- Some additional notes on the cytotaxonomy of Ruppia taxa in Western Europe.Aquatic Botany11:297‑301. https://doi.org/10.1016/0304-3770(81)90067-x
- A tetraploid cytotype of Najus marina L.Hereditas106(2):289‑291. https://doi.org/10.1111/j.1601-5223.1987.tb00262.x
- Cytotaxonomic analyses of the genus Monochoria in Asia.APG: Acta phytotaxonomica et geobotanica47:105‑111. URL: http://ci.nii.ac.jp/naid/110003758862
- Cytological study on Acorus L. in southwestern China, with some cytogeographical notes on A. calamus.Acta Botanica Sinica43(4):354‑358. [InChinese]. URL: http://www.jipb.net/pubsoft/content/2/2013/X000331(PS2).pdf
- A preliminary investigation and cytotaxonomic study on Najadaceae in China.Journal of Wuhan Botanical Research3(1):29‑44. [InChinese]. URL: http://caod.oriprobe.com/articles/826892/zhong_guo_ci_zao_ke_zhi_wu_de_chu_bu_diao_cha_ji_xi_bao_xue_fen_lei_ya.htm
- Chromosome numbers and karyotype analysis of 9 Species in Hydrocharitaceae.Acta Phytotaxonomica Sinica24(5):370‑375. URL: http://www.plantsystematics.com/qikan/epaper/zhaiyao.asp?bsid=13947
- Cytotaxonomic studies on Alisma L. from Hubei.Acta Phytotaxonomica Sinica25(4):254‑263. [InChinese]. URL: http://www.plantsystematics.com/qikan/epaper/zhaiyao.asp?bsid=14007
- A preliminary study on the polyploid series and cytogeography of Najas graminea.Acta Phytotaxonomica Sinica29(3):230‑234. [InChinese]. URL: http://www.plantsystematics.com/qikan/epaper/zhaiyao.asp?bsid=10769