|
Biodiversity Data Journal :
Forum Paper
|
|
Corresponding author: Quentin Groom (quentin.groom@plantentuinmeise.be)
Academic editor: Vincent Smith
Received: 02 Mar 2021 | Accepted: 13 May 2021 | Published: 15 Jun 2021
© 2021 Quentin Groom, Tim Adriaens, Sandro Bertolino, Kendra Phelps, Jorrit Poelen, DeeAnn Reeder, David Richardson, Nancy Simmons, Nathan Upham
This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation:
Groom Q, Adriaens T, Bertolino S, Phelps K, Poelen JH, Reeder DM, Richardson DM, Simmons NB, Upham N (2021) Holistic understanding of contemporary ecosystems requires integration of data on domesticated, captive and cultivated organisms . Biodiversity Data Journal 9: e65371. https://doi.org/10.3897/BDJ.9.e65371
|
|
Abstract
Domestic and captive animals and cultivated plants should be recognised as integral components in contemporary ecosystems. They interact with wild organisms through such mechanisms as hybridization, predation, herbivory, competition and disease transmission and, in many cases, define ecosystem properties. Nevertheless, it is widespread practice for data on domestic, captive and cultivated organisms to be excluded from biodiversity repositories, such as natural history collections. Furthermore, there is a lack of integration of data collected about biodiversity in disciplines, such as agriculture, veterinary science, epidemiology and invasion science. Discipline-specific data are often intentionally excluded from integrative databases in order to maintain the “purity” of data on natural processes. Rather than being beneficial, we argue that this practise of data exclusivity greatly limits the utility of discipline-specific data for applications ranging from agricultural pest management to invasion biology, infectious disease prevention and community ecology. This problem can be resolved by data providers using standards to indicate whether the observed organism is of wild or domestic origin and by integrating their data with other biodiversity data (e.g. in the Global Biodiversity Information Facility). Doing so will enable efforts to integrate the full panorama of biodiversity knowledge across related disciplines to tackle pressing societal questions.
Keywords
Darwin core, interoperability, invasive species, One Health, urban ecology
Introduction
Even by conservative estimates, 29% of the global land surface has been significantly modified by anthropogenic activities (
In this context, we examine the importance of integrating data on domestic and captive animals and cultivated plants by reviewing interactions with their wild counterparts. We also demonstrate how some citizen science projects reject or actively discourage observations of domestic, captive and cultivated organisms and how biodiversity data, collected by agriculture, horticulture and veterinary disciplines, are not integrated with other biodiversity datasets.
Here we briefly review the importance of data on the distributions and populations of domestic organisms for tackling some of the global ecological challenges and we make recommendations as to how the situation can be improved. We define domestic organisms as those organisms that would not exist at a particular location were it not for human intervention and where every part of their life cycle is managed, including their food, shelter, reproduction and ultimately harvesting, by humans. Despite the intense management of domestic organisms, interactions with wild organisms frequently occur and consequently play an integral role in shaping ecosystems.
Predation, parasitism and herbivory
Domestic organisms can have significant negative impacts on native biodiversity when they are allowed to roam freely. In Italy, as in many countries, domestic cats (Felis catus) predate more than 200 other species, routinely killing birds, mammals, reptiles and amphibians (
Herbivory by livestock can also have a major impact on ecosystems. Grassland covers between 12% and 21% of the global land surface and the population of cattle is close to a billion head (
The direct impacts of domestic organisms on ecosystems do not just involve mammals. Fish and shellfish are frequently stocked in natural waterways and coastal areas for recreational fishing, biocontrol or their aesthetic qualities. Introduced fish can alter natural ecosystems through interactions with native species, including increased competition and/or predation. For example, stocked brown trout (Salmo trutta) can reduce native invertebrate communities, even if those stocked fish are unable to create viable populations (
The characteristics of cultivated plants and the way that they are grown is likely to have a large influence on whether the plants have a positive or negative impact on wild organisms. For example, crop and forestry monocultures can have negative consequences for wild bees, whereas domestic gardens may provide benefits (
Genetic impacts
Hybridization between wild organisms and their domestic counterparts is widely perceived as a threat to the conservation of native biodiversity. It occurs, for example, between wild canids and domesticated dogs (
In contrast, others see the hybridization of closely related wild and domestic species brought into “artificial sympatry” not as a threat to genetic integrity, but as a mechanism whereby new biological entities are created that could, conceivably, be better suited than native species to new, human-dominated environments (
Wildlife disease
There is ample evidence for the interchange of infectious diseases between domestic animals, including livestock (
In aquatic ecosystems, aquaculture facilities not isolated from wild ecosystems have the potential to increase disease in wild fish populations. This might occur through disease spillover to wild congeners of farmed species or to other species. Captive fish populations can act as reservoirs of disease or otherwise affect disease dynamics in nearby wild populations (
Potentially invasive species
Cultivated plants, pets, wildfowl collections and aquarist collections are among the largest sources of invasive species (
Urban ecology and agroecology
Urban ecosystems and gardens are unique and interesting in their own right (
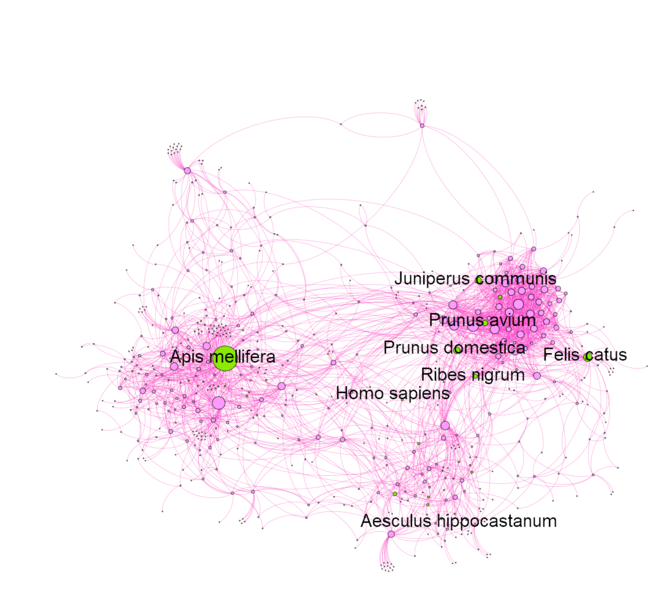
As a demonstration of the importance of domestic organisms in urban ecosystems, we constructed a species interaction network for wild and cultivated organisms recorded at Meise Botanic Garden in Belgium. Only two domesticated animals are present in the Garden, honey bees (Apis mellifera) and domestic cats from neighbouring houses (Fig.
A species interaction network of the organisms recorded for Meise Botanic Garden in Belgium. It demonstrates how the people, cultivated plants and domesticated animals (green nodes) are integrated into the ecosystem of the Garden through their interactions with wild organisms (pink nodes). Species included are only those available on GBIF (
Observations of domestic and cultivated species
Volunteers are a major contributor to ecological and biogeographic data (
“The main reason we try to mark things like this [captive/cultivated] is because iNat is primarily about observing wild organisms, not animals in zoos, garden plants, specimens in drawers, etc., and our scientific data partners are often not interested in (or downright alarmed by) observations of captive or cultivated organisms. ”
Any observation on iNaturalist marked captive/cultivated will never reach “Research Grade”. It will, therefore, not be transferred to GBIF, even if the species identification is validated. It is germane that iNaturalist puts the responsibility for this decision on their “scientific data partners”. They are not alone – eBird, the single largest contributor to GBIF, explicitly requests users not to record captive birds, escaped pets, domestic fowl and pet birds (
Other citizen science initiatives have bucked the trend and have specifically tried to survey the occurrence of alien and native plants in gardens (e.g.
The gaps in available data on domestic/captive/invasive species are plainly evident in GBIF. For example, there are approximately 26 billion chickens (Gallus gallus domesticus) in the world (
The causes and solutions
There is no doubt that all organisms, be they native, non-native, growing wild, in captivity or in cultivation, are important components of biodiversity. Suggestions on how to deal with data in these different categories have generated lively debate among biologists. For example,
Part of the reason for the artificial demarcation between wild and domestic/cultivated organisms is the divisions of research domains, industrial sectors and their respective regulatory bodies. Researchers and managers in agriculture, animal husbandry, the pet trade, epidemiology, conservation, forestry, ecology and invasion science are all interested in these data, but also generate data for their own needs. Traditionally, biodiversity observation data have been the preserve of biogeographers and conservationists and observations of cultivated and domesticated organisms are removed before creating maps and building distribution models (
For at least the past 400 years, Western culture has considered the realms of humans and nature as separate (
Another reason for observations of domesticated organisms being excluded from biodiversity datasets is that there has lacked a means by which these observations can be distinguished from those of wild organisms. The preeminent standard used to communicate biodiversity observations is the Darwin Core standard (
It is unreasonable to expect systematic observation of all domesticated organisms to be collected. Indeed, projects devoted to the study of wild organisms do not want to be swamped with large numbers of observations of pets and garden plants. However, some of these data are already collected by national and regional authorities for veterinary and agricultural statistics, pathogen surveillance and animal welfare (Table
Examples of datasets related to domestic organisms that could be incorporated into biodiversity datasets if correctly documented and standardised.
|
Sector |
Type |
Example |
|
Agriculture |
Crop map |
|
|
Livestock survey |
||
|
Aphid monitoring |
||
| Disease host specificity | ||
|
Veterinary Science |
Records of parasites, such as Hypoderma sp. (Warble fly) and Fasciola hepatica (liver fluke) |
|
|
Bees hive inspections for parasites |
||
|
Horticulture |
Inventory of botanic garden |
|
|
Observations of garden plants |
||
|
Domestic animals |
Pets census |
|
|
Zoo inventory |
|
Given that a data standard now exists (i.e.
In conclusion, although it is fairly self-evident to an ecologist that domestic organisms are part of ecosystems, data on these organisms remain poorly integrated into global data systems and are thus often disregarded. Yet, these data are highly relevant to solving many environmental challenges and should, therefore, be more actively gathered and shared.
Acknowledgements
We thank Wouter Addink, Ana Casino and Dimitrios Koureas for organising the COVID-19 Task Force of the Consortium of European Taxonomic Facilities (CETAF) and Distributed System of Scientific Collections (DiSSCo) and to other members of the Knowledge Hub group.
Funding program
QG and TA acknowledge the support of the Belgian Science Policy Office under the TrIAS project (BR/165/A1/TrIAS). QG, TA & SB acknowledge COST Action CA17122 Alien CSI Increasing understanding of alien species through citizen science, supported by COST (European Cooperation in Science and Technology). DMR acknowledges support from the DSI-NRF Centre of Excellence for Invasion Biology and the Oppenheimer Memorial Trust (grant 18576/03). SB was funded by the University of Turin, local research grant number.
Author contributions
QG & TA had the initial concept for the paper. QG wrote the outline and all authors developed the ideas and contributed to the final draft.
Conflicts of interest
The authors have no conflicts of interest to declare.
References
- Urban ecosystems: Ecological principles for the built environment.Cambridge University Press,Cambridge. https://doi.org/10.1017/CBO9780511981050
- Trying to engage the crowd in recording invasive alien species in Europe: experiences from two smartphone applications in northwest Europe.Management of Biological Invasions6(2):215‑225. https://doi.org/10.3391/mbi.2015.6.2.12
- Potential ecosystem services of urban agriculture: a review.PeerJ Preprints4:e2286v1. https://doi.org/10.7287/peerj.preprints.2286v1
- Effects of stocked trout on stream invertebrate communities.Journal of Freshwater Ecology32(1):95‑102. https://doi.org/10.1080/02705060.2016.1248502
- The roles of habitat features, disturbance, and distance from putative source populations in structuring alien plant invasions at the urban/wildland interface on the Cape Peninsula, South Africa.Biological Conservation132(2):183‑198. https://doi.org/10.1016/j.biocon.2006.03.023
- Agroecología urbana: diseño de granjas urbanas ricas en biodiversidad, productivas y resilientes.Agro Sur46(2):49‑60. https://doi.org/10.4206/agrosur.2018.v46n2-07
- Connecting to nature through tech? The case of the iNaturalist app.Convergence: The International Journal of Research into New Media Technologies27(1):124‑141. https://doi.org/10.1177/1354856520933064
- One health: Its origins and future. In:One health: The human-animal-environment interfaces in emerging infectious diseases.365.Springer Berlin Heidelberg,Berlin,13pp. [ISBN978-3-642-36889-9]. https://doi.org/10.1007/82_2012_223
- Four-legged friend or foe? Dog walking displaces native birds from natural areas.Biology Letters3(6):611‑613. https://doi.org/10.1098/rsbl.2007.0374
- The biomass distribution on Earth.Proceedings of the National Academy of Sciences115(25):6506‑6511. https://doi.org/10.1073/pnas.1711842115
- A framework for prioritising present and potentially invasive mammal species for a national list.NeoBiota62:31‑54. https://doi.org/10.3897/neobiota.62.52934
- A proposed unified framework for biological invasions.Trends in Ecology & Evolution26(7):333‑339. https://doi.org/10.1016/j.tree.2011.03.023
- Spillover of functionally important organisms between managed and natural habitats.Agriculture, Ecosystems & Environment146(1):34‑43. https://doi.org/10.1016/j.agee.2011.09.005
- Gene flow from domesticated escapes alters the life history of wild Atlantic salmon.Nature Ecology & Evolution1(5). https://doi.org/10.1038/s41559-017-0124
- Temporal change in genetic integrity suggests loss of local adaptation in a wild Atlantic salmon (Salmo salar) population following introgression by farmed escapees.Heredity106(3):500‑510. https://doi.org/10.1038/hdy.2010.165
- Collateral diseases: Aquaculture impacts on wildlife infections.Journal of Applied Ecologyhttps://doi.org/10.1111/1365-2664.13775
- Contribution of citizen science towards international biodiversity monitoring.Biological Conservation213:280‑294. https://doi.org/10.1016/j.biocon.2016.09.004
- Crop feeding by brown howlers (Alouatta guariba clamitans) in forest fragments: The conservation value of cultivated species.International Journal of Primatology38(2):263‑281. https://doi.org/10.1007/s10764-016-9927-8
- Anthropogenic food subsidies hinder the ecological role of wolves: Insights for conservation of apex predators in human-modified landscapes.Global Ecology and Conservation21https://doi.org/10.1016/j.gecco.2019.e00841
- Parasite spread at the domestic animal - wildlife interface: anthropogenic habitat use, phylogeny and body mass drive risk of cat and dog flea (Ctenocephalides spp.) infestation in wild mammals.Parasites & Vectors11(1). https://doi.org/10.1186/s13071-017-2564-z
- Working with gardeners to identify potential invasive ornamental garden plants: testing a citizen science approach.Biological Invasions20(11):3069‑3077. https://doi.org/10.1007/s10530-018-1759-3
- Relative population densities and predation of the common vampire bat (Desmodus rotundus) in natural and cattle-raising areas in north-east Argentina.Preventive Veterinary Medicine14(1-2):13‑20. https://doi.org/10.1016/0167-5877(92)90080-Y
- Urban vegetable for food security in cities. A review.Agronomy for Sustainable Development35(2):483‑498. https://doi.org/10.1007/s13593-014-0273-y
- Anthropogenic transformation of the terrestrial biosphere.Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences369(1938):1010‑1035. https://doi.org/10.1098/rsta.2010.0331
- Anthropogenic Biomes: 10,000 BCE to 2015 CE.Land9(5). https://doi.org/10.3390/land9050129
- Hybridization as a stimulus for the evolution of invasiveness in plants?Proceedings of the National Academy of Sciences97(13):7043‑7050. https://doi.org/10.1073/pnas.97.13.7043
- Arboreta reveal the invasive potential of several conifer species in the temperate forests of western Europe.NeoBiota64:23‑42. https://doi.org/10.3897/neobiota.64.56027
- The food and agriculture organization FAOSTAT. http://www.fao.org/faostat/en/#data. Accessed on: 2021-2-06.
- Biodiversity quality: a paradigm for biodiversity.Ecological Indicators10(6):1077‑1082. https://doi.org/10.1016/j.ecolind.2010.04.002
- Virulent Newcastle disease viruses from chicken origin are more pathogenic and transmissible to chickens than viruses normally maintained in wild birds.Veterinary Microbiology235:25‑34. https://doi.org/10.1016/j.vetmic.2019.06.004
- A review of mutual transmission of important infectious diseases between livestock and ildlife in Europe.Annals of the New York Academy of Sciences969(1):4‑13. https://doi.org/10.1111/j.1749-6632.2002.tb04343.x
- The aquarium and horticultural industry as a pathway for the introduction of aquatic invasive species—outreach initiatives within the Great Lakes basin.Biodiversity10(2-3):104‑112. https://doi.org/10.1080/14888386.2009.9712852
- Non-native species in urban environments: Patterns, processes, impacts and challenges.Biological Invasions19:3461‑3469. https://doi.org/10.1007/s10530-017-1598-7
- Predation by wolves (Canis lupus) on wild and domestic ungulates of the western Alps, Italy.Journal of Zoology266(2):205‑213. https://doi.org/10.1017/S0952836905006801
- Occurrence Download.The Global Biodiversity Information Facility. https://doi.org/10.15468/DL.BGF9GB
- Impacts of stocking and its intensity on effective population size in brook charr (Salvelinus fontinalis) populations.Conservation Genetics20(4):729‑742. https://doi.org/10.1007/s10592-019-01168-2
- Do managed bees drive parasite spread and emergence in wild bees?International Journal for Parasitology: Parasites and Wildlife5(1):64‑75. https://doi.org/10.1016/j.ijppaw.2015.10.001
- Leishmaniases of the New World: current concepts and implications for future research.Clinical Microbiology Reviews6(3):230‑250. https://doi.org/10.1128/CMR.6.3.230
- Improving Darwin Core for research and management of alien species.Biodiversity Information Science and Standards3https://doi.org/10.3897/biss.3.38084
- AgentschapPlantentuinMeise/gardenNetwork: submission (Version 0.1.4).0.1.4.Zenodo. Release date:2021-1-29. URL: https://doi.org/10.5281/zenodo.4573274
- Quantifying the value of user-level data cleaning for big data: A case study using mammal distribution models.Ecological Informatics34:139‑145. https://doi.org/10.1016/j.ecoinf.2016.06.001
- Domestic dogs and cats as sources of Trypanosoma cruzi infection in rural northwestern Argentina.Parasitology134(1):69‑82. https://doi.org/10.1017/S0031182006001259
- The population dynamics of an alien insect: Andricus quercuscalicis (Hymenoptera: Cynipidae).The Journal of Animal Ecology60(2). https://doi.org/10.2307/5297
- Urban biodiversity: comparison of insect assemblages on native and non-native trees.Urban Ecosystems15(3):611‑624. https://doi.org/10.1007/s11252-012-0231-x
- Managing the whole landscape: historical, hybrid, and novel ecosystems.Frontiers in Ecology and the Environment12(10):557‑564. https://doi.org/10.1890/130300
- Domestic dogs (Canis familiaris) as a predator and disturbance agent of wildlife in Tasmania.Australasian Journal of Environmental Management21(4):441‑452. https://doi.org/10.1080/14486563.2014.952787
- No PhDs needed: how citizen science is transforming research.Nature562(7728):480‑482. https://doi.org/10.1038/d41586-018-07106-5
- Managing biodiversity in agricultural ecosystems.Columbia University Press, ,492pp. https://doi.org/10.1007/s10745-009-9280-z
- Urban gardens promote bee foraging over natural habitats and plantations.Ecology and Evolution6(5):1304‑1316. https://doi.org/10.1002/ece3.1941
- Hybridization between escaped domestic and wild American mink (Neovison vison).Molecular Ecology18(6):1175‑1186. https://doi.org/10.1111/j.1365-294X.2009.04100.x
- Establishment risk from pet-trade freshwater turtles in the European Union.Knowledge and Management of Aquatic Ecosystemshttps://doi.org/10.1051/kmae/2013057
- Exploring reservoir dynamics: a case study of rabies in the Serengeti ecosystem.Journal of Applied Ecology45(4):1246‑1257. https://doi.org/10.1111/j.1365-2664.2008.01468.x
- Impact of hybridization with domestic dogs on the conservation of wild canids.Free-ranging dogs and wildlife conservation170https://doi.org/10.1093/acprof:osobl/9780199663217.003.0007
- A review of cat behavior in relation to disease risk and management options.Applied Animal Behaviour Science173:29‑39. https://doi.org/10.1016/j.applanim.2015.07.002
- Domestic dogs (Canis familiaris) as reservoir hosts for Rickettsia conorii.Vector-Borne and Zoonotic Diseases12(1):28‑33. https://doi.org/10.1089/vbz.2011.0684
- The pet trade as a source of invasive fish in Taiwan.Taiwania51(2):93‑98. https://doi.org/10.6165/tai.2006.51(2).93
- When pets become pests: the role of the exotic pet trade in producing invasive vertebrate animals.Frontiers in Ecology and the Environment17(6):323‑330. https://doi.org/10.1002/fee.2059
- Crop domestication and the disruption of species interactions.Basic and Applied Ecology11(2):116‑125. https://doi.org/10.1016/j.baae.2009.11.008
- A survey of the transmission of infectious diseases/infections between wild and domestic ungulates in Europe.Veterinary Research42(1). https://doi.org/10.1186/1297-9716-42-70
- Bats track and exploit changes in insect pest populations.PLOS One7(8). https://doi.org/10.1371/journal.pone.0043839
- Ecology of domestic dogs Canis familiaris as an emerging reservoir of Guinea worm Dracunculus medinensis infection.PLOS Neglected Tropical Diseases14(4). https://doi.org/10.1371/journal.pntd.0008170
- Small urban centres as launching sites for plant invasions in natural areas: insights from South Africa.Biological Invasions19(12):3541‑3555. https://doi.org/10.1007/s10530-017-1600-4
- Interaction effects of different drivers of wild bee decline and their influence on host–pathogen dynamics.Current Opinion in Insect Science26:136‑141. https://doi.org/10.1016/j.cois.2018.02.007
- Vespa velutina: a new invasive predator of honeybees in Europe.Journal of Pest Science87(1):1‑16. https://doi.org/10.1007/s10340-013-0537-3
- License to kill? Domestic cats affect a wide range of native fauna in a highly biodiverse Mediterranean country.Frontiers in Ecology and Evolution7:477. https://doi.org/10.3389/fevo.2019.00477
- Domestic dogs are mammalian reservoirs for the emerging zoonosis flea-borne spotted fever, caused by Rickettsia felis.Scientific Reports10(1). https://doi.org/10.1038/s41598-020-61122-y
- Invasive plant species and the ornamental horticulture industry. In:Management of Invasive Weeds.5.Springer Netherlands,Dordrecht. [ISBN978-1-4020-9201-5 978-1-4020-9202-2]. https://doi.org/10.1007/978-1-4020-9202-2_9
- Ethics for wildlife conservation: Overcoming the human–nature dualism.BioScience56(2). https://doi.org/10.1641/0006-3568(2006)056[0144:EFWCOT]2.0.CO;2
- Biodiversity assessments: Origin matters.PLOS Biology16(11). https://doi.org/10.1371/journal.pbio.2006686
- Dark side of the fence: ornamental plants as a source of wild-growing flora in the Czech Republic.Preslia88(2).
- Global biotic interactions: Interpreted data products.Zenodo.Version Number: 0.2 type: dataset DOI: 10.5281/ZENODO.3950589. URL: https://zenodo.org/record/3950589
- Global biotic interactions: An open infrastructure to share and analyze species-interaction datasets.Ecological Informatics24:148‑159. https://doi.org/10.1016/j.ecoinf.2014.08.005
- Quantifying the contribution of citizen science to broad‐scale ecological databases.Frontiers in Ecology and the Environment18(1):19‑26. https://doi.org/10.1002/fee.2128
- Integrated behavioural and stable isotope data reveal altered diet linked to low breeding success in urban-dwelling blue tits (Cyanistes caeruleus).Scientific Reports7(1). https://doi.org/10.1038/s41598-017-04575-y
- Does vegetation structure influence criminal activity? Insights from Cape Town, South Africa.Frontiers of Biogeography11(1). https://doi.org/10.21425/F5FBG42035
- Agricultural intensification, priming for persistence and the emergence of Nipah virus: a lethal bat-borne zoonosis.Journal of The Royal Society Interface9(66):89‑101. https://doi.org/10.1098/rsif.2011.0223
- Spillover edge effects: the dispersal of agriculturally subsidized insect natural enemies into adjacent natural habitats.Ecology Letters9(5):603‑614. https://doi.org/10.1111/j.1461-0248.2006.00911.x
- Global livestock production systems.Food and Agriculture Organization of the United Nations (FAO) and International Livestock Research Institute (ILRI),Rome,152pp. URL: http://www.fao.org/3/i2414e/i2414e00.htm [ISBN978-92-5-107028-4]
- Wild pollinator activity negatively related to honey bee colony densities in urban context.PLOS One14(9). https://doi.org/10.1371/journal.pone.0222316
- Gardens benefit bees and enhance pollination in intensively managed farmland.Biological Conservation144(11):2602‑2606. https://doi.org/10.1016/j.biocon.2011.07.008
- Ecological diversity of birds in relation to the structure of urban green space.Landscape and Urban Planning77(1-2):39‑53. https://doi.org/10.1016/j.landurbplan.2005.01.004
- Dogs are reservoir hosts for possible transmission of human strongyloidiasis in Thailand: molecular identification and genetic diversity of causative parasite species.Journal of Helminthology94https://doi.org/10.1017/S0022149X1900107X
- Do non-native species contribute to biodiversity?PLOS Biology16(4). https://doi.org/10.1371/journal.pbio.2005568
- Quantifying the contributions of native and non-native trees to a city’s biodiversity and ecosystem services.Urban Forestry & Urban Greening56https://doi.org/10.1016/j.ufug.2020.126861
- Wildlife presence and interactions with chickens on Australian commercial chicken farms assessed by camera traps.Avian Diseases62(1):65‑72. https://doi.org/10.1637/11761-101917-Reg.1
- Hybridization between wild and cultivated potato species in the Peruvian Andes and biosafety implications for deployment of GM potatoes.Euphytica164(3):881‑892. https://doi.org/10.1007/s10681-007-9641-x
- eBird: A citizen-based bird observation network in the biological sciences.Biological Conservation142(10):2282‑2292. https://doi.org/10.1016/j.biocon.2009.05.006
- Global change and local solutions: Tapping the unrealized potential of citizen science for biodiversity research.Biological Conservation181:236‑244. https://doi.org/10.1016/j.biocon.2014.10.021
- The Anthropocene could raise biological diversity.Nature502(7469):7‑7. https://doi.org/10.1038/502007a
- The changing role of ornamental horticulture in alien plant invasions: Horticulture and plant invasions.Biological Reviews93(3):1421‑1437. https://doi.org/10.1111/brv.12402
- How well do we understand the impacts of alien species on ecosystem services? A pan-European, cross-taxa assessment.Frontiers in Ecology and the Environment8(3):135‑144. https://doi.org/10.1890/080083
- Bescherming en beheer in Vlaanderen.Mededelingen van het Instituut voor Natuuren Bosonderzoek2:79‑88. [Inn]. URL: https://www.vlaanderen.be/publicaties/natuurrapport-2020
- Prediction and attenuation of seasonal spillover of parasites between wild and domestic ungulates in an arid mixed-use system.Journal of Applied Ecology55(4):1976‑1986. https://doi.org/10.1111/1365-2664.13083
- Where and what do we record?https://bsbi.org/wp-content/uploads/dlm_uploads/Where_and_what_do_we_record.pdf. Accessed on: 2020-12-27.
- Global spread of helminth parasites at the human-domestic animal-wildlife interface.Global Change Biology24(7):3254‑3265. https://doi.org/10.1111/gcb.14064
- Dogs as agents of disturbance. In:Free-ranging dogs and wildlife onservation.Oxford University Press,Oxford,336pp. URL: https://global.oup.com/academic/product/free-ranging-dogs-and-wildlife-conservation-9780199663217?cc=nl&lang=en& [ISBN9780199663217].
- Darwin Core: An evolving community-developed biodiversity data standard.PLOS One7(1). https://doi.org/10.1371/journal.pone.0029715
- The FAIR Guiding Principles for scientific data management and stewardship.Scientific Data3(1). https://doi.org/10.1038/sdata.2016.18
- Can urban greening increase vector abundance in cities? The impact of mowing, local vegetation, and landscape composition on adult mosquito populations.Urban Ecosystems22(5):827‑839. https://doi.org/10.1007/s11252-019-00857-7